

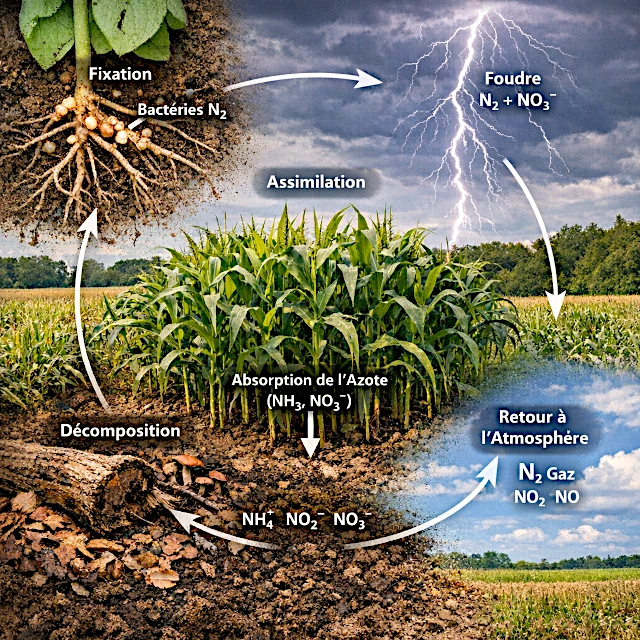
Parce que l’azote, indispensable à la construction des protéines et de l’ADN, ne peut être utilisé directement par la plupart des êtres vivants sous sa forme gazeuse pourtant abondante dans l’atmosphère. Le cycle de l’azote transforme continuellement cet élément, grâce à l’action combinée des bactéries, des plantes, des animaux et des phénomènes naturels, en formes assimilables par le vivant. Sans ces conversions successives — fixation, nitrification, assimilation, ammonification et dénitrification — les écosystèmes s’effondreraient rapidement. Ce cycle invisible mais fondamental maintient l’équilibre biologique de la planète et conditionne la fertilité des sols, la productivité des océans et la stabilité du climat.
Earth's atmosphere contains nearly 78% dinitrogen (\(N_2\)) by volume or 75% by mass. Yet, this vast potential resource for life is inaccessible to most living beings. No animal, human, or plant can transform it into nutrients.
The reason lies in the exceptional strength of the triple chemical bond between the two nitrogen atoms (N≡N). Considerable energy is required to break this bond.
Two pathways exist: lightning at 30,000 °C, or the chemical elegance of bacteria and their nitrogenase enzyme, capable of reducing \(N_2\) to \(NH_3\) (dinitrogen → ammonia). Without this prior rupture, there would be no proteins, no DNA, no muscles, no neurons, no blood: no life.
The nitrogen cycle is a cascade of transformations that converts an inert gas into assimilable nutrients, then returns it to the atmosphere.
N.B.: Antoine Lavoisier (1743-1794) proposed the term "nitrogen" (from Greek "a-zôê", without life). Paradoxically, this inert element in gaseous form proves indispensable to every living cell once fixed.
| Process | Chemical transformation | Bacteria + Fungi | Required conditions | Ecological role |
|---|---|---|---|---|
| Symbiotic fixation | \(N_2 \rightarrow NH_3\) | Rhizobium, Bradyrhizobium | Symbiosis with legume roots, microaerophilia | Direct nitrogen supply to cultivated plants |
| Free fixation | \(N_2 \rightarrow NH_3\) | Azotobacter (aerobic), Clostridium (anaerobic) | Soils, aquatic environments, available organic carbon | Diffuse but constant contribution to natural fertility |
| Nitrification (step 1) | \(NH_3 \rightarrow NO_2^-\) | Nitrosomonas, Nitrosospira | Strict aerobiosis, neutral to slightly alkaline pH | Formation of nitrites, precursors of nitrates |
| Nitrification (step 2) | \(NO_2^- \rightarrow NO_3^-\) | Nitrobacter, Nitrospira | Aerobiosis, tolerance to a wide range of temperatures | Production of nitrates, highly assimilable form for plants |
| Denitrification | \(NO_3^- \rightarrow N_2\) (via \(NO_2^-\), NO, \(N_2O\)) | Pseudomonas, Paracoccus, Bacillus | Anoxia (saturated soils, sediments), labile organic matter | Cycle closure, return of \(N_2\) to the atmosphere |
| Ammonification (mineralization) | Organic N \(\rightarrow NH_4^+\) | Saprophytic fungi (Bacillus, Streptomyces, etc.) | Aerobiosis or anaerobiosis, decomposition of necromass | Recycling of nitrogen from organic waste |
N.B.: Human activity through synthetic fertilizers has doubled the flow of reactive nitrogen entering terrestrial ecosystems since 1950.
Before the industrial era, natural fluxes of biological and atmospheric fixation maintained a stable stock of reactive nitrogen in soils and waters. The work of Justus von Liebig (1803-1873) had already foreseen the importance of nitrogen compounds for fertility. But it was the invention of the Haber-Bosch process (first decade of the 20th century) that multiplied the production of synthetic nitrogen fertilizers. Today, anthropogenic fixation exceeds natural fixation.
Excess nitrates leached by rain reach rivers and then coastal areas, causing eutrophic dead zones where algal blooms suffocate aquatic fauna. The Gulf of Mexico or the Baltic Sea illustrate this diffuse pollution. Moreover, nitrous oxide (\(N_2O\)), a byproduct of nitrification and denitrification, is a greenhouse gas 300 times more potent than carbon dioxide, contributing to global warming.
It is the set of processes that convert atmospheric nitrogen into biologically usable forms and eventually return it to the atmosphere.
Because the N₂ molecule is extremely stable and only certain bacteria can break it apart.
They drive the key steps: nitrogen fixation, nitrification, ammonification, and denitrification, making nitrogen available or returning it to the air.
They absorb nitrates and ammonium from the soil to build proteins, amino acids, and DNA.
Fertilizers, pollution, and industrial emissions add excess reactive nitrogen, causing eutrophication, ecological imbalance, and greenhouse gas release.