

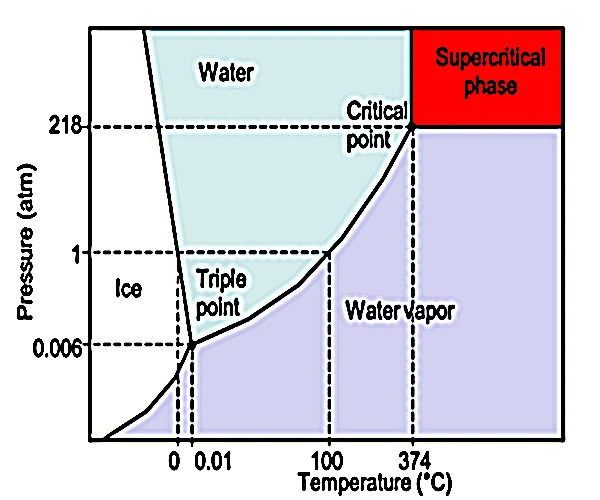
The supercritical state of water, reached at a temperature of 374 °C and a pressure of 22.06 MPa (≈217.8 atm), is a thermodynamic domain in which water no longer distinguishes between liquid and gas. In this zone, it has both the density of a liquid and the diffusivity of a gas. This hybrid behavior leads to unique physical and chemical properties, making it highly reactive, excellent for dissolving organic and inorganic substances, and widely used in geochemistry, industrial processes, and even in the exploration of prebiotic conditions.
Unlike liquid water or steam, supercritical water exhibits significant density fluctuations on a small scale, which alters dielectric constants, ionic solvation, and thermal transport mechanisms. It can dissolve salts like a liquid but behave like a gas for non-polar species. Its thermal conductivity is high, its viscosity low, and its surface tension is zero. These characteristics make it an excellent working fluid or green solvent in sustainable chemistry processes.
The supercritical state of water can only exist at pressures greater than 217 atm and temperatures above 374 °C. These conditions are naturally found in certain deep geothermal environments, particularly in the oceanic crust and near magma chambers. It can be found in the following areas:
These extreme environments are of major interest both for fundamental research (origin of life, extremophiles, mineralization) and for emerging geoenergy technologies.
Supercritical water is found in various fields:
The supercritical state of water may have played a role in the origin of life on Earth. In deep hydrothermal environments, supercritical water may have favored complex organic reactions, precursors to the appearance of the first biological molecules. Furthermore, the exceptional solvation and catalysis properties may have supported the development of prebiotic chemistries on the surface or in the Earth's crust.
| Property | Liquid | Vapor | Supercritical |
|---|---|---|---|
| Density (kg/m³) | 1000 | 0.6 | 200-500 |
| Viscosity (μPa·s) | 890 | 13 | 100-300 |
| Dielectric constant | ≈80 | ≈1.0 | ≈10-20 |
| Salt solubility | Strong | None | Medium to strong (variable) |
| Surface tension | 72 mN/m | None | None |
Source: NIST Chemistry WebBook, ScienceDirect - Supercritical water oxidation.