

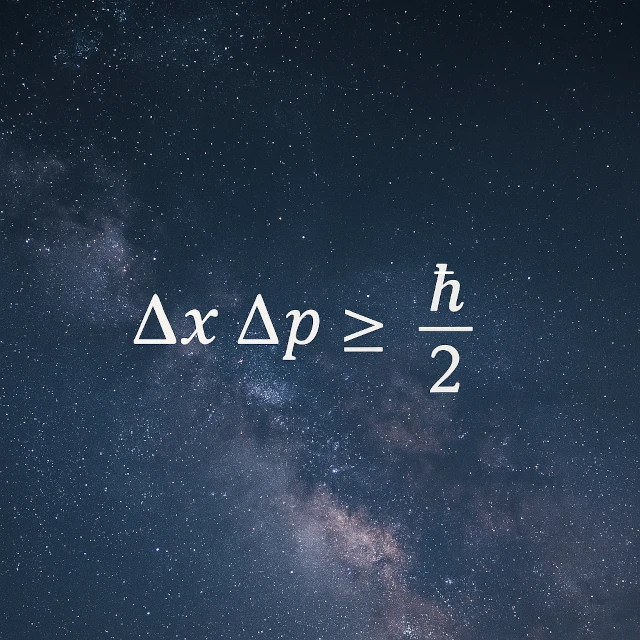
Heisenberg's uncertainty principle (Δx × Δp ≥ ħ/2) states that it is impossible to simultaneously know both the position (Δx) and momentum (Δp) of a quantum particle with precision. The more precisely you know the position, the less you know its speed, and vice versa. If a particle were stationary, it would mean its position (fixed) and momentum (zero) were both perfectly determined, violating this inequality. At the quantum scale (e.g., for an electron in an atom), confining a particle to a small space automatically gives it motion: stationarity is therefore impossible.
Einsenberg's formula is a fundamental discovery of quantum mechanics. Formulated in 1927 by Werner Einsenberg (1901-1976), it expresses a fundamental limit to the precision with which certain pairs of physical properties, such as position (x) and momentum (p), can be simultaneously known.
This relationship is mathematically expressed by the inequality: Δx * Δp ≥ ħ/2, where \(\hbar = \frac{h}{2\pi}\) (h = Planck's constant and ħ = reduced Planck's constant). The inequality (≥) means that the product of the two uncertainties (Δx) and (Δp) can never fall below ħ/2.
In other words: The more precisely the position of a particle (Δx) is measured, the less precisely its momentum (Δp) can be known, and vice versa. This is a fundamental limit of nature, not a technical limitation of our instruments. We can never know both the position and the velocity of a quantum particle simultaneously with absolute precision.
Δx (delta x) represents the uncertainty in the position of a particle. It is the "margin of error" or imprecision with which we know exactly where the particle is in space. Δp (delta p) represents the uncertainty in the momentum (or impulse) of the particle. The momentum p = mv (mass × velocity), so Δp measures the imprecision in the speed/movement of the particle. The product Δx * Δp therefore represents the product of these two uncertainties.
ħ (h-bar) is the reduced Planck constant, equal to h/2π, where h is Planck's constant (approximately 1.054 × 10⁻³⁴ J·s). It is a fundamental constant of nature that characterizes the quantum scale. ħ/2 is therefore half of this constant.
The smallness of ħ does not mean that at our macroscopic scale, we "know" the position and movement of an object. It means that our usual measurement inaccuracies are enormous compared to ħ/2, thus masking the quantum effects of macroscopic objects. The inequality is always widely verified in practice.
At the quantum scale, our measurement precisions are on the order of 10⁻¹⁵ m, far from ħ/2. What changes is that the compromise imposed by ħ/2 becomes relevant for understanding the behavior of particles. The uncertainty principle becomes physically significant at this scale because confining an electron in a space of 10⁻¹⁰ m (the size of an atom) automatically implies that it has movement, and reducing Δx requires increasing Δp according to the relation ΔxΔp ≥ ħ/2.
An electron that is both immobile and localized is therefore impossible, which explains why no particle can be at rest.
Imagine you are trying to take a very sharp photo of a car moving at high speed. To get a sharp image, you must use a very short exposure time. However, a short exposure time means you capture less light, which can make the image dark or blurry if the light is insufficient.
Δx (delta x) represents the uncertainty in the particle's position. Δp (delta p) represents the uncertainty in its momentum (mass × velocity). Their product can never be less than ħ/2, where ħ is the reduced Planck constant (about 1.054 × 10⁻³⁴ J·s). This limit is fundamental: it does not come from a flaw in our instruments, but from the intrinsic nature of the quantum world.
Yes, but its effects are completely imperceptible at our scale. The constant ħ is extremely small. For a macroscopic object, usual measurement imprecisions are enormous compared to ħ/2, so the inequality is always easily verified. It is only at the quantum scale (atoms, electrons, photons) that the trade-off imposed by ħ/2 becomes physically significant and determines particle behavior.
It has several major consequences: it shows that quantum reality is intrinsically probabilistic (replacing classical determinism). It explains energy quantization in atoms and molecules. It is fundamental for quantum tunneling (a particle crossing an energy barrier classically insurmountable), used in tunnel transistors and microscopes. It implies that the quantum vacuum is never perfectly empty but subject to fluctuations, and it is intimately linked to wave-particle duality.