

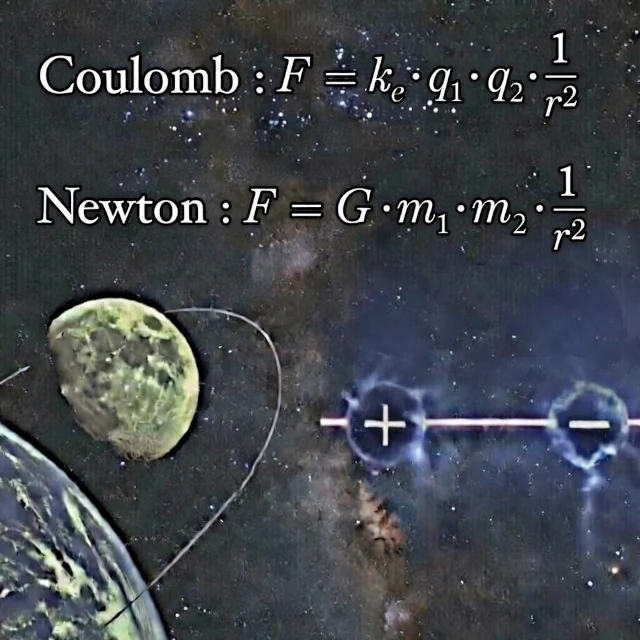
Why does the Moon orbit the Earth? Why does your hair stand on end after removing a wool hat? The answer to these two seemingly unrelated questions is written in almost identical mathematical structures. On one hand, the law of universal gravitation formulated by Isaac Newton (1643-1727) at the end of the 17th century. On the other, the law of electrostatics established by Charles-Augustin de Coulomb (1736-1806) at the end of the 18th century. Both are "inverse-square laws of distance".
For gravity: \( F_g = G \cdot \frac{m_1 \cdot m_2}{r^2} \)
For electricity: \( F_e = k_e \cdot \frac{q_1 \cdot q_2}{r^2} \)
In these equations, \( F \) represents force, \( m \) mass, \( q \) electric charge, and \( r \) the distance between the two objects. The constants \( G \) and \( k_e \) are merely scaling factors. Is this striking similarity a cosmic coincidence or a sign of a deeper truth about the structure of the Universe?
The most fundamental reason for this similarity is geometric.
We live in a three-dimensional space. Imagine a point source of influence, whether it is a mass or a charge. This influence spreads spherically and isotropically (identical in all directions) around it.
At a distance \( r \) from the source, all the emitted influence must pass through an imaginary spherical surface. Why a surface and not a volume? Because the influence spreads like a spherical wavefront. At any given moment, all points located at the same distance \( r \) from the source are reached simultaneously. These points form precisely a spherical surface, not a volume.
The surface area of a sphere is given by the formula \( 4\pi r^2 \). The energy or intensity of the effect (called flux) is therefore uniformly distributed over this surface as it moves away from the source.
If the source emits a total amount of influence per second that we call \( Q \) (a flow rate), then the intensity \( I \) measured at a point located at distance \( r \) is this total amount divided by the surface over which it spreads: \( I = \frac{Q}{4\pi r^2} \)
This expression means that the further away you are, the larger the surface to be crossed (\( 4\pi r^2 \) increases with \( r \)), and thus the lower the intensity per unit area. It is exactly like the light of a candle: at 1 meter, it illuminates a certain surface; at 2 meters, the same amount of light must illuminate a surface 4 times larger (since \( (2)^2 = 4 \)), so the light intensity per unit area is 4 times weaker.
Volume does not come into play here because we measure an effect at a point on the surface, not in a three-dimensional space. The relevant physical quantity is the surface density of flux, not a volumetric density.
This expression breaks down into two parts:
- \( Q \) is a constant that depends only on the source (its emission power).
- \( \frac{1}{4\pi r^2} \) is a geometric factor that describes how the influence is diluted over a spherical surface.
Mathematically, we can rewrite: \( I = \left( \frac{Q}{4\pi} \right) \times \frac{1}{r^2} \)
The term \( \frac{Q}{4\pi} \) is a global constant for a given source. The dependence on distance is therefore entirely contained in the factor \( \frac{1}{r^2} \). This is why we say that intensity is inversely proportional to the square of the distance.
In Coulomb's and Newton's laws, we find this same structure:
- For Coulomb: \( F = k_e \cdot q_1 \cdot q_2 \cdot \frac{1}{r^2} \)
- For Newton: \( F = G \cdot m_1 \cdot m_2 \cdot \frac{1}{r^2} \)
The constants \( k_e \) and \( G \) already integrate geometric factors such as \( \frac{1}{4\pi} \) (in the appropriate unit system, such as the rationalized SI system), as well as the fundamental properties of the interaction.
If we lived in a two-dimensional universe, the influence would be distributed over a circle (a perimeter) of length \( 2\pi r \), and the law would become \( I = \frac{Q}{2\pi r} = \left( \frac{Q}{2\pi} \right) \times \frac{1}{r} \), i.e., a \( 1/r \) law. In four dimensions, it would follow a \( 1/r^3 \) law. The inverse-square law is therefore the mathematical signature of a force propagating in a three-dimensional space.
While the mathematical form is identical, the deep nature of these two forces is radically different. The first difference is obvious: mass is always positive, so gravity is always attractive. Electric charges, on the other hand, can be positive or negative. The electric interaction can therefore be both attractive and repulsive.
Thus, Coulomb's law has two complementary faces that structure our reality:
It is this duality that makes Coulomb's law one of the most elegant and fundamental pillars of physics. Without it, the Universe would be nothing more than an undifferentiated soup of particles, without structure, without life, and without a table to rest your hand on.
The second difference lies in intensity. The electric force is colossally more powerful than the gravitational force. To convince yourself, compare the electric repulsion force between two protons with their gravitational attraction force. The ratio is about \( 10^{36} \) in favor of electricity. If gravity dominates at the scale of planets and galaxies, it is only because matter is electrically neutral on a large scale. Positive and negative charges cancel each other out, leaving only the weak but omnipresent gravity.
| Property | Gravitation (Newton) | Electrostatics (Coulomb) |
|---|---|---|
| Source | Mass (always positive) | Electric charge (positive or negative) |
| Nature of the force | Always attractive | Attractive or repulsive |
| Constant | \( G = 6.674 \times 10^{-11} \, \text{N·m}^2/\text{kg}^2 \) | \( k_e = 8.988 \times 10^9 \, \text{N·m}^2/\text{C}^2 \) |
| Relative intensity | \( 10^{-36} \) (the weakest) | \( 10^{36} \) (much stronger) |
| Screening | Impossible (no negative mass) | Possible (electric neutrality) |
| Domain of predominance | Astronomical (planets, galaxies) | Atomic and molecular |
This resemblance has fascinated the greatest minds. Albert Einstein (1879-1955) spent the last thirty years of his life attempting to unify gravity and electromagnetism into a single theory, a "unified field theory." He failed, but his quest paved the way for modern physics. Today, we know that these two forces are low-energy manifestations of deeper theories.
Gravity is described by general relativity as a curvature of spacetime. Electromagnetism, of which electrostatics is a part, is described by quantum electrodynamics. The unification of these two pillars (general relativity and quantum mechanics) remains the Holy Grail of theoretical physics, a quest to understand why, at a fundamental level, these two forces, so different, share this elegant mathematical structure.